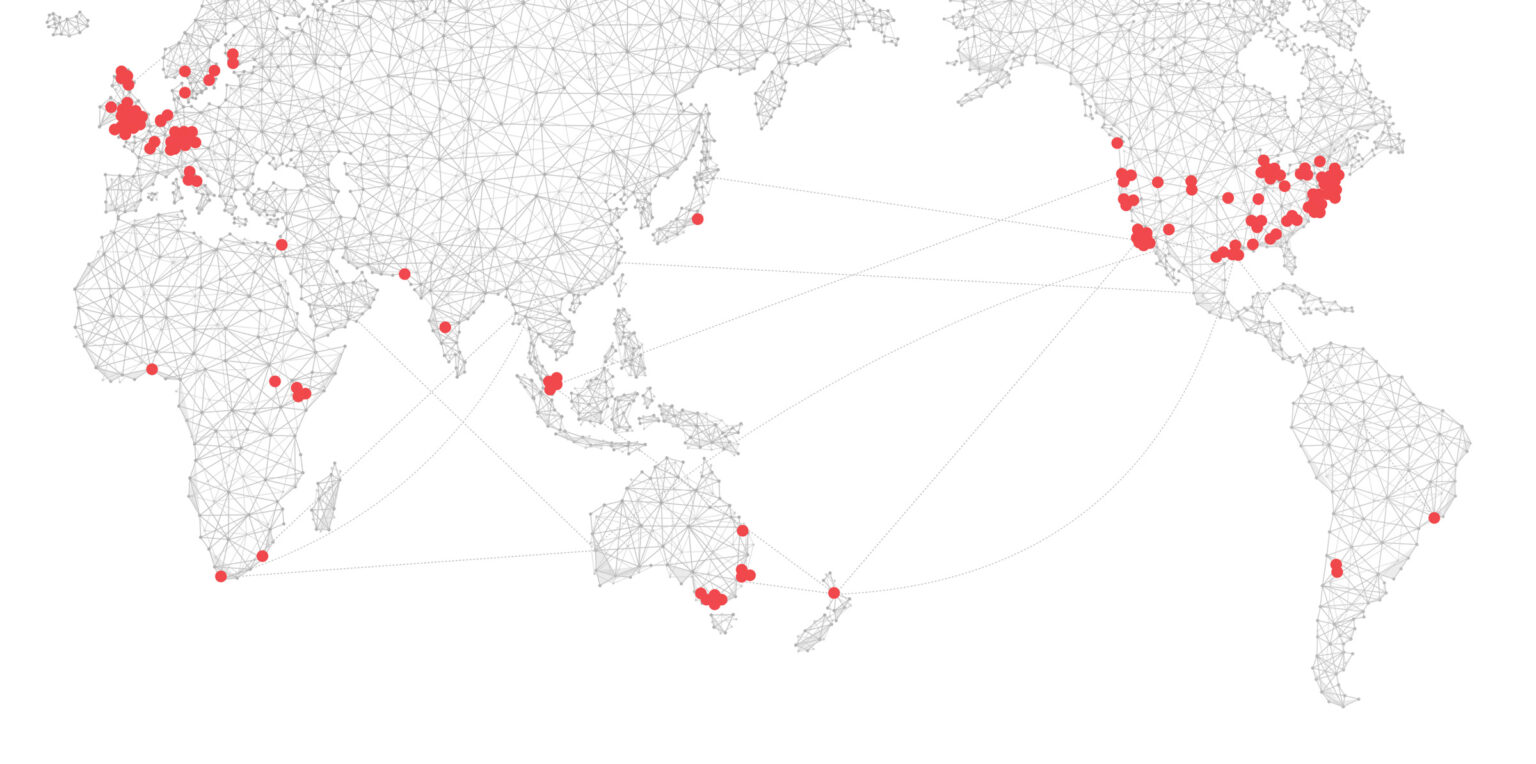
A global network dedicated to accelerating breakthroughs in human health.
Now over 170 organizations on 6 continents, representing more than 1,500,000 researchers.
The world’s largest research network, ready for instant activation.
The Wellcome Leap Health Breakthrough Network is the largest, most rapidly ‘activatable’ health research network in the world. With more than 170 world-class institutions, non-profits and commercial entities representing a network of over 1,500,000 scientists and engineers across six continents, we are able to increase speed and collaboration, reducing barriers to breakthroughs in human health.
The Wellcome Leap Health Breakthrough Network will help eliminate barriers to progress thanks to the first-of-its-kind master research funding agreement, which equitably addresses all terms and conditions, including IP, ownership, and publication. Once an organization signs the agreement, any researcher or group will need to negotiate only the statement of work and cost before funds can be transmitted and work can begin. Often in days, shaving months or more off of development timelines.
Network Members
- Across Platforms UG
- Africa Health Research Institute (AHRI)
- The Aga Khan University
- Agency for Science, Technology and Research (A*STAR)
- Algorithmiq Inc.
- Arizona State University
- Baker Heart and Diabetes Institute
- Baylor College of Medicine
- Ben-Gurion University of the Negev
- Boston Children’s Hospital
- Boston University
- The Brigham & Women’s Hospital
- Burnet Institute
- California Institute of Technology
- Cardiff University
- Carnegie Mellon University
- Central Institute of Mental Health Mannheim
- Centillion Technology Ltd.
- ChipSense Inc.
- City of Hope
- The Cleveland Clinic Foundation
- Cornell University
- Dana-Farber Cancer Institute
- Deakin University
- Design-Zyme LLC
- DNA Script
- Duke University
- Emory University
- Fondazione Toscana Life Sciences
- Francis Crick Institute
- Frontier Bio Corporation
- Georgia Tech
- German BioImaging
- Glencoe Software, Inc.
- Grendel Games
- Harvard John A. Paulson School of Engineering and Applied Sciences
- Harvard Medical School
- Helmholtz Zentrum München
- Huntsman Mental Health Institute at the University of Utah
- Icahn School of Medicine at Mount Sinai
- Imperial College London
- Indian Institute of Science Bangalore
- Indiana University
- Infleqtion Inc.
- Karolinska Institutet
- King’s College London
- Lariox Healthcare Ltd
- Lattice Automation
- Loughborough University
- Louisiana State University
- Makerere University
- Massachusetts General Hospital
- Massachusetts Institute of Technology
- Max Planck Society
- McLean Hospital
- Monash University
- Nanyang Technological University
- National University of Singapore
- New Hope Molecular, LLC
- NMI Natural and Medical Sciences Institute
- Northeastern University
- Pixelgen Technologies
- Princeton University
- qBraid co.
- RareCyte, Inc.
- Revalia Bio Inc.
- Rice University
- RIKEN
- The Rosalind Franklin Institute
- Rutgers
- RiboPro
- Sibel Health Inc.
- SIMPL: Society for Improving Medical Professional Learning
- Soihtu DTx
- Surgical Safety Technologies
- Sixfold Bioscience
- Stanford University
- Strand Therapeutics Inc.
- Swansea University
- Trestle Biotherapeutics
- Tufts University
- Universidad de Chile
- University College London
- University of Auckland
- University of Birmingham
- University of British Columbia
- University of California, Irvine
- University of California, Los Angeles
- University of California San Diego
- University of Cambridge
- University of Cape Town
- University of Colorado Boulder
- University of Copenhagen
- University of Dundee
- The University of Edinburgh
- University of Helsinki
- University of Lagos
- University of Leeds
- The University of Manchester
- University of Massachusetts Amherst
- University of Melbourne
- University of Michigan
- University of Minnesota
- The University of New South Wales
- The University of North Carolina at Charlotte
- University of Nottingham
- University of Oxford
- University of Pennsylvania
- University of Pittsburgh
- The University of Queensland
- University of São Paulo
- The University of Sheffield
- University of Southern California
- University of Strathclyde
- University of Sydney
- University of Tennessee, Knoxville
- University of Technology Sydney
- The University of Texas at Austin
- The University of Texas Health Science Center at Houston
- The University of Texas Health Science Center at San Antonio
- University of Trento
- University of Washington
- The University of York
- University of Zurich
- Uppsala University
- Vanderbilt University
- VETeRNA
- Virginia Tech
- Walter and Eliza Hall Institute of Medical Research (WEHI)
- Washington University in St. Louis
- Wayne State University
- Wellesley College
- Wellcome Sanger Institute
- Wyss Center for Bio and Neuroengineering
- Wyss Institute for Biologically Inspired Engineering at Harvard University
- Yale University
FAQs
How can I join the Wellcome Leap Health Breakthrough Network?
Institutions, non-profits and commercial entities interested in joining should send an email to HBNet@wellcomeleap.org.
When was the Wellcome Leap Health Breakthrough Network established?
It was established in January 2021 with a charter group of 21 world-class institutions. Original press release here.
What is the master research funding agreement?
If you wish to participate in Wellcome Leap programs or be a part of the Health Breakthrough Network (“HBNet”), it will be necessary for your organization to become a signatory to either Master Academic Research Funding Agreement (MARFA) or Commercial Research Funding Agreement (CORFA). The agreements contain all the basic terms and conditions governing execution of a project and receipt of funding. The MARFA/CORFA may be — and often is — signed before specific projects of interest are identified, accelerating project starts and funding for any researcher in the organization.
Because all the terms have already been agreed upon, once a proposal from an organization in the HBNet is accepted, only a Statement of Work (“SOW”) and cost need be negotiated and signed by the Performer and Wellcome Leap for work to begin. The SOW may be part of a larger program of research funded by Wellcome Leap, having a common goal (a “Wellcome Leap Program”), or an independent effort, as discussed further below.
How is my confidential information treated?
Under the MARFA/CORFA, once an effort is underway, the Performer and Wellcome Leap may exchange confidential information during the period of the funded project, subject to the standard confidentiality provisions of the MARFA/CORFA. Any information of an obviously confidential nature is protected – with or without explicit marking – consistent with our view that a strict marking requirement can limit collaborative exchange or lead to unintended consequences, i.e., “gotchas”. Performers are always free to mark their own confidential information, should they elect to do so.
Is third party funding permitted?
There is no restriction on third party funding unless that funding directly supports a Wellcome Leap-funded project. Direct third party funding of Wellcome Leap-funded projects requires our prior written approval, to ensure that there are no changes to Wellcome Leap-funded project objectives, IP ownership of any research results is clear, and that commercialization (as appropriate) is not unduly hindered.
Is there any possibility of joint invention and if so, how is ownership handled?
In theory, a joint invention could result from a Performer’s interactions with Wellcome Leap or with other Performers within the same Program. Specifically, the latter could occur during occasional group meetings organized by us to discuss progress against the Program goal. It is at the discretion of each organization to decide if and when to enter into any appropriate agreement (e.g., a mutual NDA) with the organization(s) the other Performer(s) represent.
Under the MARFA/CORFA, Wellcome Leap assigns any interest we may have in joint intellectual property to your organization. We retain a non-exclusive license solely for R&D purposes, which can be sublicensed to other non-profit entities only to ensure continued development. However, if you can show that granting this license would create an unintended hinderance to the Program goals, such as scaling and distribution, Wellcome Leap will reconsider this requirement.
Does Wellcome Leap require rights to existing intellectual property?
We require that each Performer identify any existing (background) intellectual property likely to be required for the Wellcome Leap-funded research or the potential commercialization of a resulting product or technology. To the extent your organization is able to grant a non-exclusive R&D license to us (i.e., this is not prevented by any existing agreement), we generally require that. However, as above, if the Performer can show that granting this license would create an unintended hinderance to the Program goals, Wellcome Leap will reconsider this requirement.
Does Wellcome Leap have the right to delay or prevent publication of the results?
We prioritize publication of research results, generally, and, in particular, under an open access policy. We do require each Performer to provide an advance copy of any proposed publication or public presentation in order to review the same for Wellcome Leap confidential information or potentially patentable subject matter. This review period is limited to 30 days but we will endeavor to review as quickly as possible. As you may realize, this is common in research funding agreements and we believe it is a reasonable compromise to ensure against the loss of IP rights that may be necessary to commercialize Wellcome Leap-funded research and to protect legitimate interest in any confidential information shared with a Performer. If patentable subject matter is identified, we may request a further delay until a patent application can be filed but in no case, more than an additional 30 days.
What obligation, if any, does the Performer have to commercialize the results?
Wellcome Leap anticipates that it will normally further our mission (and the organization’s mission) to commercialize the results of Wellcome Leap-funded research. If we determine that a Performer’s organization is not making appropriate efforts to further commercialization, either itself or through a third party (e.g., a licensee), we have the option to request a meeting and a remedial commercialization plan to address the issue. If, within six (6) months of the remedial plan, appropriate efforts to commercialization have not been made, Wellcome Leap reserves the right to assume responsibility for commercialization (i.e., “step-in”), either itself or through a third party. In the unlikely event we were to exercise this step-in right, it remains possible, although not guaranteed a priori, that a Performer’s organization would participate financially in the successful commercialization of the research thereafter.
Please note that Wellcome Leap views exercise of its step-in rights as an extraordinary remedy, and would not intend, nor have any motivation, to exercise this right under any but the most unusual circumstance.
Under what circumstances can the parties terminate?
Either party can terminate the MARFA/CORFA for an uncured breach and other conventional grounds (e.g., bankruptcy), as would be expected. Wellcome Leap may also terminate the MARFA/CORFA upon thirty (30) days’ prior written notice to an organization. Please note that we would not intend, nor have any motivation, to exercise this right under any but the most unusual circumstance. An organization may also terminate the MARFA/CORFA if there are no pending SOWs at a given time.
Wellcome Leap may also terminate a SOW for convenience, although would not intend, nor have any motivation, to exercise this right under normal circumstances. To preserve the integrity of integrated programs and mutual dependencies across disciplines and organizations, an organization may not terminate an ongoing SOW for convenience. Either the Performer organization or we may terminate a SOW if it is determined, in good faith, that the R&D is no longer academically, technically or commercially feasible, upon thirty (30) days’ prior written notice to the other party.
What are the eligible costs for Wellcome Leap Programs?
Full direct and government-certified indirect costs appropriate to the execution of the research, are eligible.